The synthesis of 3–octyne is achieved by adding a bromoalkane into a mixture of sodium amide and an alkyne. The bromoalkane and alkyne respectively are
BrCH2CH2CH2CH2CH3 and CH3CH2C≡CH
BrCH2CH2CH3 and CH3CH2CH2C≡CH
BrCH2CH2CH2CH2CH3 and CH3C≡CH
BrCH2CH2CH2CH3 and CH3CH2C≡CH
Additional Questions
|
Alkyl halides react with dialkyl copper reagents to give |
Answer |
|
The compound formed as a result of oxidation of ethyl benzene by KMnO4 is |
Answer |
|
Which of the following compounds will not undergo Friedel–Craft's reaction easily? |
Answer |
|
Which of the following reagents when heated with ethyl chloride, forms ethylene? |
Answer |
|
Among the following select the alkane that is expected to have lowest boiling point |
Answer |
|
Addition of HBr to propylene in presence of benzoylperoxide, follows : |
Answer |
|
The radical
|
Answer |
|
Which of the following has highest knocking? |
Answer |
|
Which one of the following methods is neither meant for the synthesis nor for separation of amines? |
Answer |
|
A petroleum fraction having boiling range 70 – 200°C and containing 6 – 10 carbon atoms per molecule is called |
Answer |

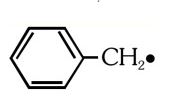 is aromatic because it has
is aromatic because it has