An organic compound X is oxidized by using acidified K2Cr2O7. The product obtained reacts with phenyl hydrazine but does not answer silver mirror test. The possible structure of X is
(CH3)2CHOH
CH3CHO
CH3CH2OH
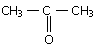

An organic compound X is oxidized by using acidified K2Cr2O7. The product obtained reacts with phenyl hydrazine but does not answer silver mirror test. The possible structure of X is
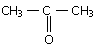
CH3CH=CHCH3 $\xrightarrow{O_3} \text{A} \underset{Zn}{\xrightarrow{H_2O}} \text{B}$
The compound B is