21703.Among the following compounds the one that is most reactive towards electrophilic nitration is
Toluene
Benzene
Benzoic Acid
Nitrobenzene
21704.What would be the product formed when 1–Bromo–3–chlorocyclobutane reacts with two equivalents of metallic sodium in ether?
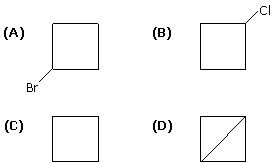

Option (A)
Option (B)
Option (C)
Option (D)
21706.Aqueous solution of an organic compound, 'A' on electrolysis liberates acetylene and CO2 at anode. 'A' is
potassium citrate
potassium succinate
potassium acetate
potassium maleate
21707.The correct statement is
Cyclohexadiene and cyclohexene cannot be isolated with ease during controlled hydrogenation of benzene.
One mole each of benzene and hydrogen when reacted gives 1/3 mole of cyclohexane and 2/3 mole unreacted hydrogen.
Hydrogenation of benzene to cyclohexane is an endothermic process.
It is easier to hydrogenate benzene when compared to cyclohexene.
21708.Pure methane can be produced by
Soda lime decarboxylation
Kolbe's electrolytic method
Wurtz reaction
Reduction with H2
21709.The first fraction obtained during the fractionation of petroleum is:
Gasoline
Diesel oil
Hydrocarbon gases
Kerosene oil
21710.Presence of a nitro group in a benzene ring
activates the ring towards electrophilic substitution
renders the ring basic
deactivates the ring towards nucleophilic substitution
deactivates the ring towards electrophilic substitution
21711.The reagents to carry out the following conversion are
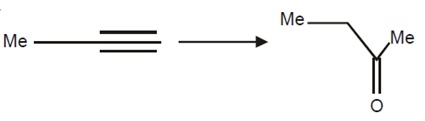
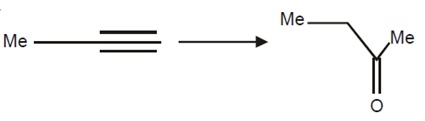
HgSO4/dil. H2SO4
BH3; H2O2/NaOH
OsO4; HIO4
NaNH2/CH3I; HgSO4/dil H2SO4
21712.The IUPAC name of the following molecule is
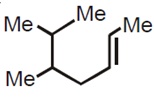

5,6–Dimethyl hept–2–ene
2,3–Dimethyl hept–5–ene
5,6–Dimethyl hept–3–ene
5–Isopropyl hex–2–ene
