The correct order of strengths of the carboxylic acids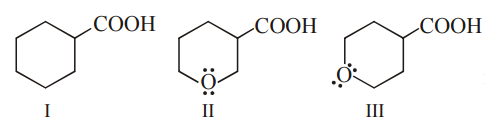
is
(II > III > I)
Acidic strength α –I, –M effect
–I effect depend upon distance so II have stronger –I effect than III.

The correct order of strengths of the carboxylic acids
is
Acidic strength α –I, –M effect
–I effect depend upon distance so II have stronger –I effect than III.
|
The compound that will react most readily with gaseous bromine has the formula: |
Answer |
|
Which one of the following compounds shows the presence of intramolecular hydrogen bond? |
Answer |
|
The molar conductivity of a 0.5 mol / dm3 solution of AgNO3 with electrolytic conductivity of 5.76 × 10-3 S cm-1 at 298 K is |
Answer |
|
The decomposition of phosphine (PH3) on tungsten at low pressure is a first-order reaction. It is because the |
Answer |
|
The coagulation values in millimoles per litre of the electrolytes used for the coagulation of As2S3 are given below: |
Answer |
|
During the electrolysis of molten sodium chloride, the time required to produce 0.10 mol of chlorine gas using a current of 3 amperes is |
Answer |
|
How many electrons can fit in the orbital for which n = 3 and l = 1 ? |
Answer |
|
For a sample of perfect gas when its pressure is changed isothermally from pi to pf, the entropy change is given by |
Answer |
|
The vant Hoff factor (i) for a dilute aqueous solution of the strong electrolyte barium hydroxide is |
Answer |
|
The percentage of pyridine (C5H5N) that forms pyridinium ion (C5H5N+H) in a 0.10 M aqueous pyridine solution (Kb for C5H5N = 1.7 × 10–9) is |
Answer |