Consider the reaction
CH3CH2CH2Br + NaCN → CH3CH2CH2CN + NaBr
This reaction will be the fastest in
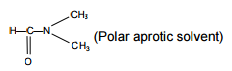
CH3CH2CH2Br + NaCN → CH3CH2CH2CN + NaBr
This is a SN2 Reaction for which polar aprotic medium is suitable for faster rate of reaction.

Consider the reaction
CH3CH2CH2Br + NaCN → CH3CH2CH2CN + NaBr
This reaction will be the fastest in
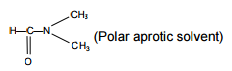
CH3CH2CH2Br + NaCN → CH3CH2CH2CN + NaBr
This is a SN2 Reaction for which polar aprotic medium is suitable for faster rate of reaction.
|
The correct structure of the product A formed in the reaction |
Answer |
|
Which among the given molecules can exhibit tautomerism? |
Answer |
|
The correct order of strengths of the carboxylic acids |
Answer |
|
The compound that will react most readily with gaseous bromine has the formula: |
Answer |
|
Which one of the following compounds shows the presence of intramolecular hydrogen bond? |
Answer |
|
The molar conductivity of a 0.5 mol / dm3 solution of AgNO3 with electrolytic conductivity of 5.76 × 10-3 S cm-1 at 298 K is |
Answer |
|
The decomposition of phosphine (PH3) on tungsten at low pressure is a first-order reaction. It is because the |
Answer |
|
The coagulation values in millimoles per litre of the electrolytes used for the coagulation of As2S3 are given below: |
Answer |
|
During the electrolysis of molten sodium chloride, the time required to produce 0.10 mol of chlorine gas using a current of 3 amperes is |
Answer |
|
How many electrons can fit in the orbital for which n = 3 and l = 1 ? |
Answer |